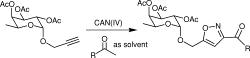Publications
48) Bahadori, S.; Leclerc, C.; St-Gelais, J.; Poulin, G.; Giguère, D. Catalyst-Controlled, Site-Selective Functionalization of Levoglucosan Analogues Mediated by Chiral Zinc Diamine Complexes. J. Org. Chem. 2025, 90, 1940-1949.
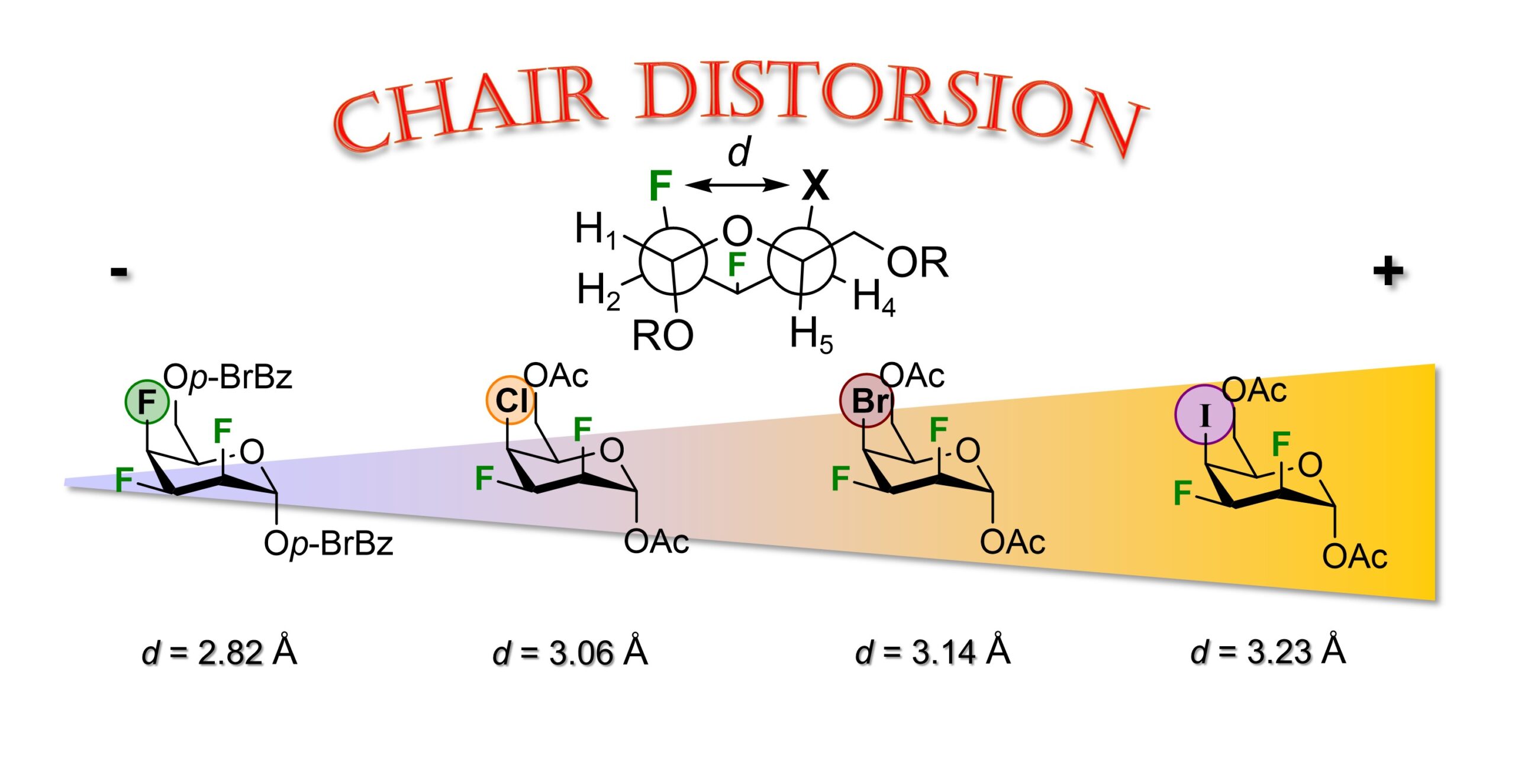
47) Lessard, O.; Grosset-Magagne, M.; Johnson, P. A.; Giguère, D. Synthesis and Conformational Analysis of Pyran Inter-Halide Analogues of D-Talose. Beilstein J. Org. 2024, 20, 2442-2454. Part of the thematic issue « Organo-fluorine chemistry VI », Guest Editor: D. O’Hagan.
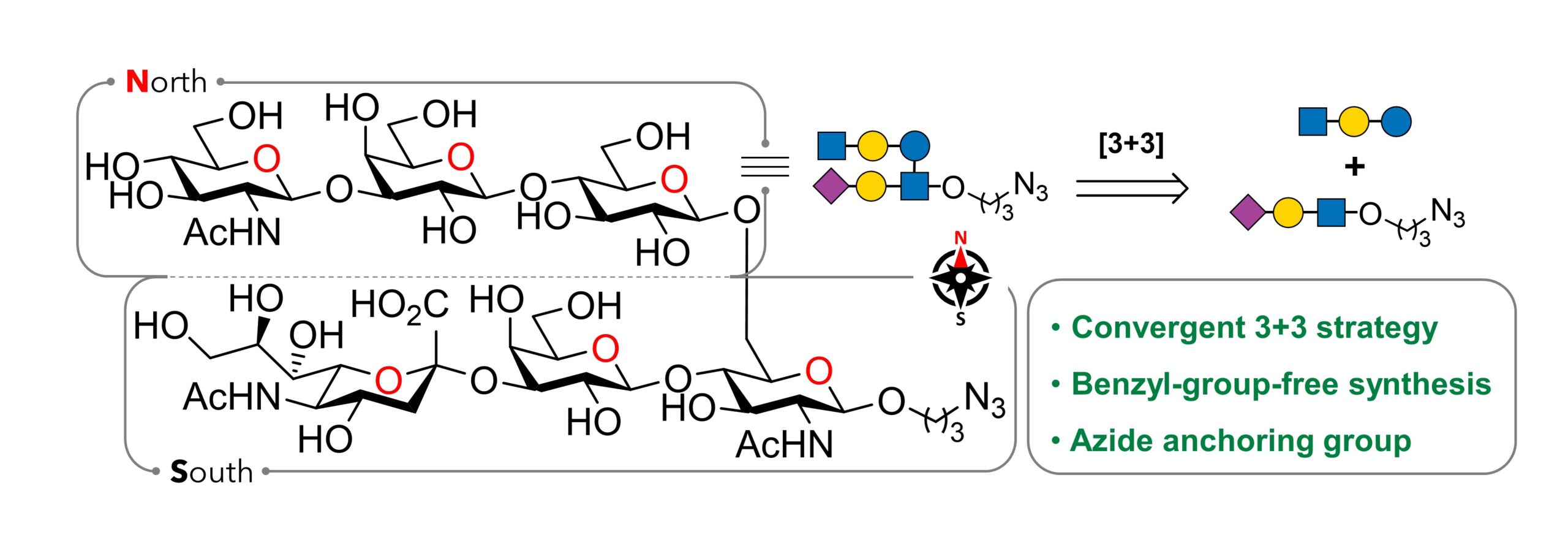
46) Bahadori, S.; Archambault, M.-J.; Sebastio, M.; Bourgault, S.; Giguère, D. Convergent Synthesis of a Group B Stretococcus Type III Epitope Towards a Semisynthetic Carbohydrates-Based Vaccine. J. Org. Chem. 2024, 89, 13978-13992.
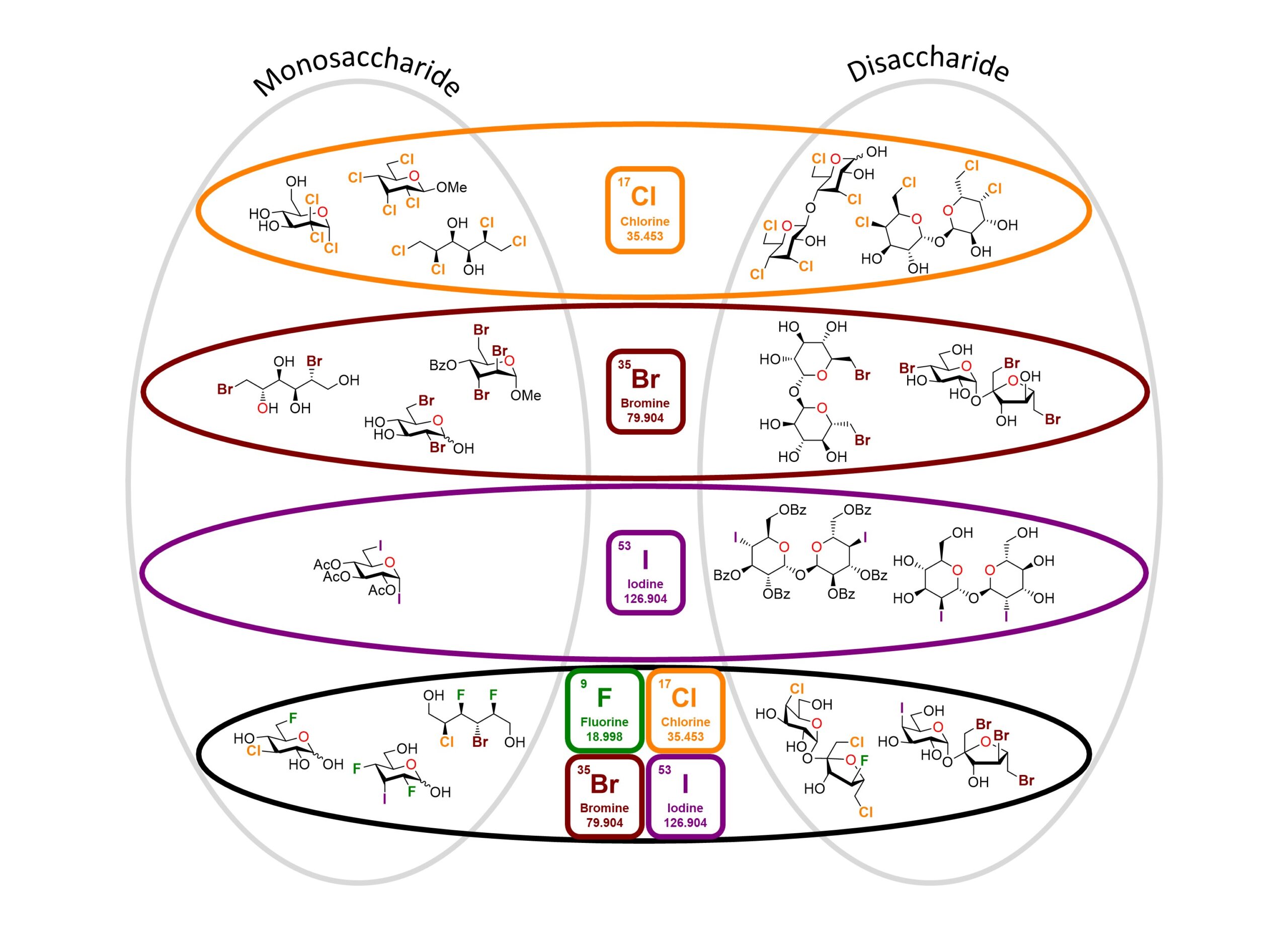
45) Lessard, O.; Lainé, D.; Giguère, D. Polyhalogenated Carbohydrates: Synthesis and Applications of Sugar Halides from Fluorine to Iodine. Eur. J. Org. Chem. 2024, 27, e202400120.
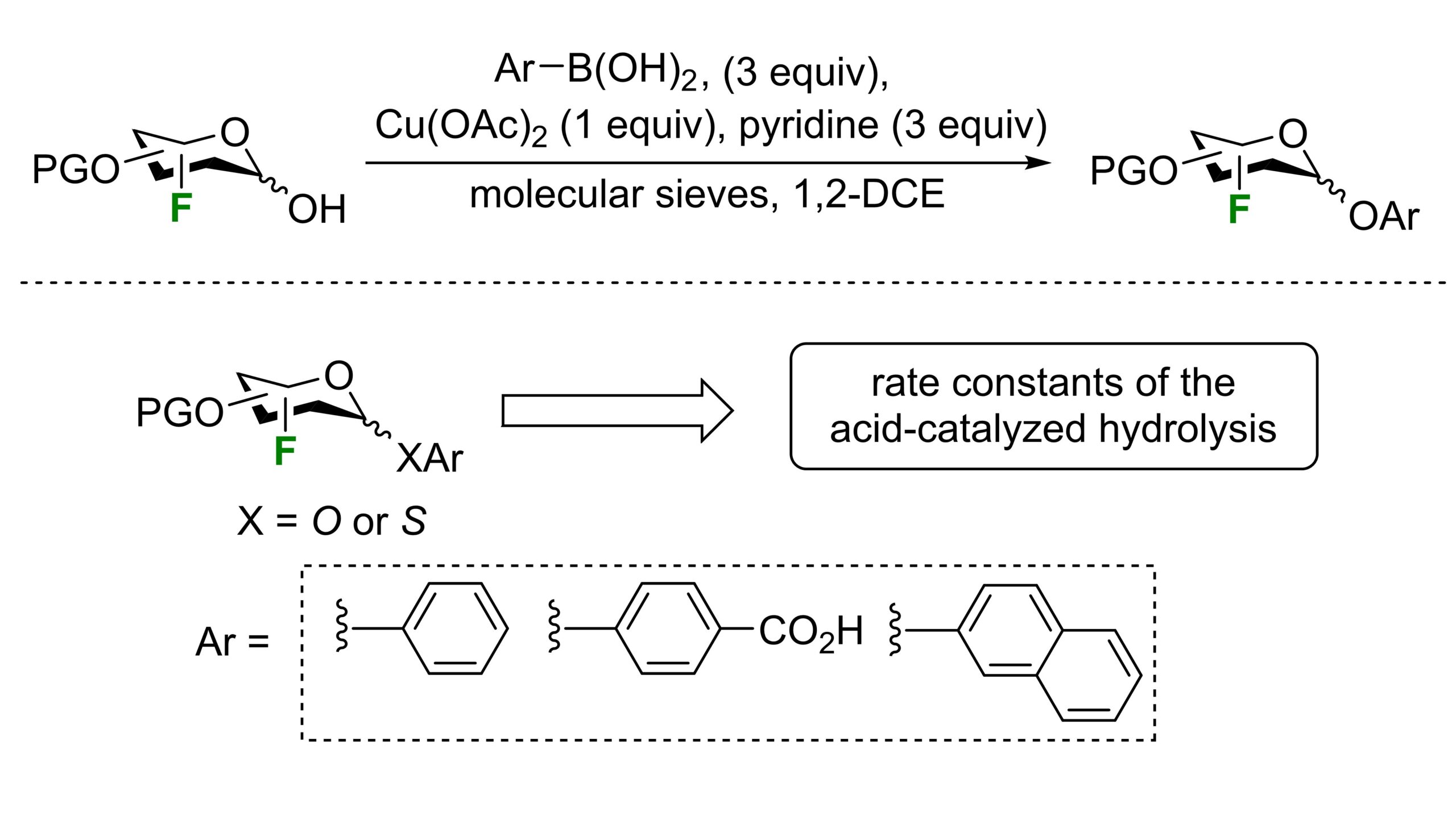
44) Leclerc, C.; St-Gelais, J.; Cecioni, S.; Giguère, D. Synthesis and hydrolysis of aryl fluoroglycosides. J. Fluorine Chem. 2024, 273, 110232.
43) Bricha, S.; Cote-Cyr, M.; Tremblay, T.; Nguyen, P. T.; St-Louis, P.; Giguère, D.; Archambault, D.; Bourgault, S. Synthetic multicomponent nanovaccines based on the molecular co-assembly of β-peptides protect against influenza A virus. ACS Infect. Dis. 2023, 9, 1232-1244.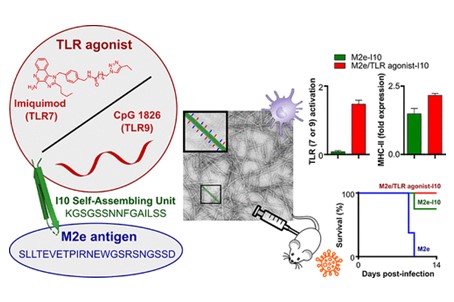
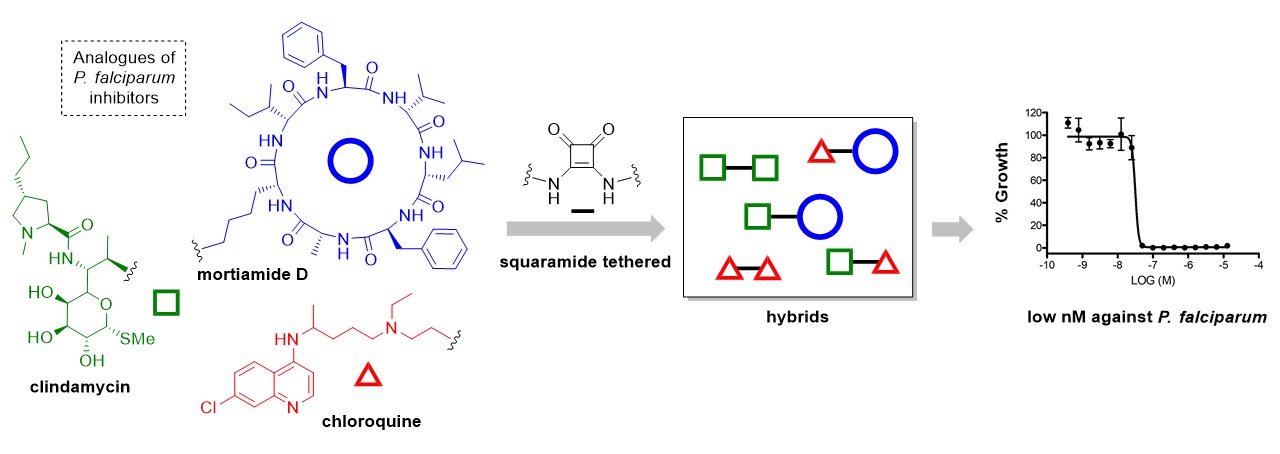
42) Tremblay, T.; Bergeron, C.; Gagnon, D.; Bérubé, C.; Voyer, N.; Richard, D.; Giguère, D. Squaramide tethered clindamycin, chloroquine, and mortiamide hybrides: Design, synthesis, and antimalarial activity. ACS Med. Chem. Lett. 2023, 14, 217-222.
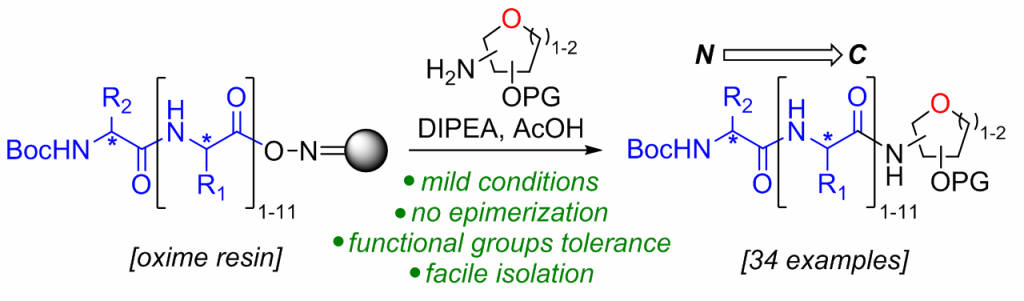
41) Tremblay, T.; Haguette, P.; Robert-Scott, G.; Alcée, J. B.; Bérubé, C.; Bergeron, C.; Voyer, N.; Giguère, D. Synthesis of Lincosamide Analogues via Oxime Resin Aminolysis. Synlett, 2023, 34, 40-44.
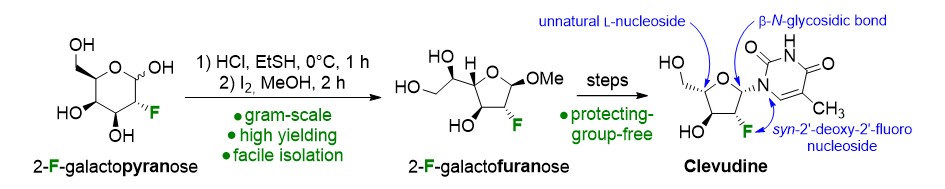
40) Tremblay, T.; Alcée, J. B.; Giguère, D. Protecting-group-free synthesis of clevudine (L-FMAU), a treatment of the hepatitis B virus. Org. Biomol. Chem. 2022, 20, 8859-8863.
39) Lessard, O.; Lainé, D.; Fecteau, C.-É.; Johnson, P. A.; Giguère, D. Fundamental Curiosity of Multivicinal Inter-Halide Stereocenters. Org. Chem. Front. 2022, 9, 6566-6572.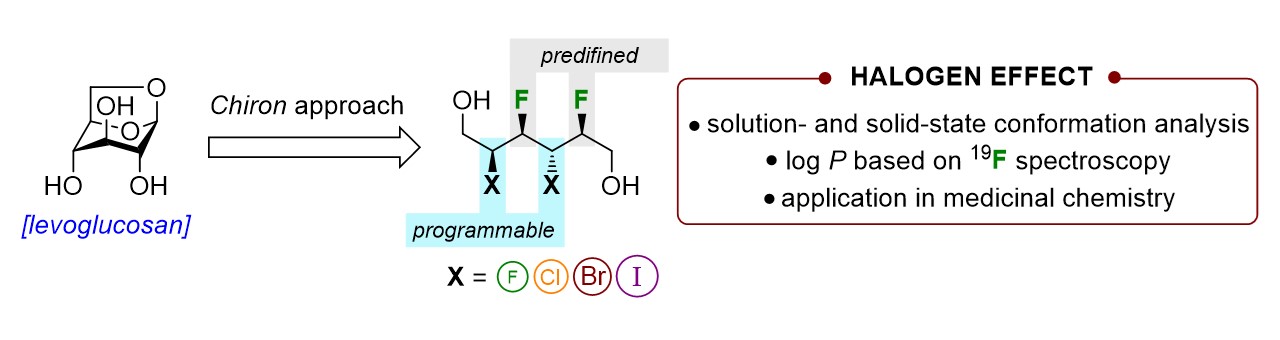
38) St-Gelais, J.; Leclerc, C.; Giguère, D. Synthesis of fluorinated thiodigalactoside analogues. Carbohydr. Res. 2022, 511, 108481.
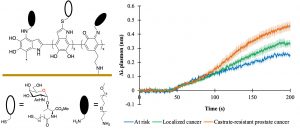
37) Bouchard, M.; Tremblay, T.; Paré-Lacroix, M.-P.; Gagné-Boulet, M.; Fortin, S.; Giguère, D. Synthetic development of sugar amino acid oligomers towards novel podophyllotoxin analogues. Bioorg. Med. Chem. 2021, 52, 116501.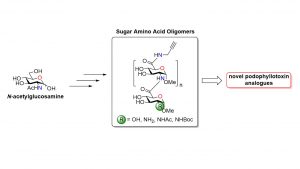 36) Lamarre, M.; Tremblay, T.; Bansept, M.-A.; Robitaille, K.; Fradet, V.; Giguère, D.; Boudreau, D. A glycan-based plasmonic sensor for prostate cancer diagnosis. Analyst 2021, 146, 6852-6860.
36) Lamarre, M.; Tremblay, T.; Bansept, M.-A.; Robitaille, K.; Fradet, V.; Giguère, D.; Boudreau, D. A glycan-based plasmonic sensor for prostate cancer diagnosis. Analyst 2021, 146, 6852-6860.
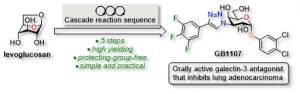
35) St-Gelais, G.; Giguère, D. Protecting-Group-Free Synthesis of GB1107, an Orally Active Galectin-3 Antagonist. Synthesis, 2021, 53, 3735-3743. Feature Article.
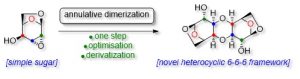
34) Robert-Scott, G.; St-Gelais, J.; Giguère, D. Annulative Dimerization of Carbohydrates: Synthesis of complex C2-Symmetrical 1,4-Dioxane-Sugar Hybrids. Eur. J. Org. Chem. 2021, 22, 3322-3330.
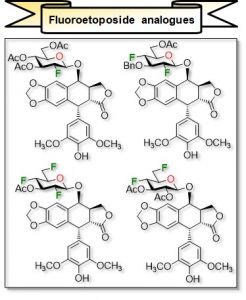
33) Tremblay, T.; St-Gelais, J.; Houde, M.; Giguère, D. Polyfluoroglycoside Synthesis via Simple Alkylation of an Anomeric Hydroxyl Groups: Access to Fluoroetoposide Analogues. J. Org. Chem. 2021, 86, 4812-4824.
32) Lainé, D.; Lessard, O.; St-Gelais, J.; Giguère, D. From carbohydrates to complex organofluorine: Synthesis, conformation, and lipophilicity of Multi-Vicinal Fluorin-Containing hexitol analogues. Chem. Eur. J. 2021, 27, 3799-3805.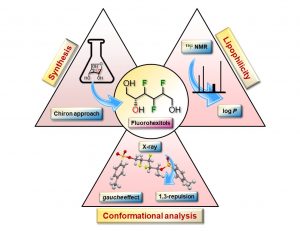
31) Lainé, D.; Denavit, V.; Lessard, O.; Carrier, L.; Fecteau, C.-É.; Johnson, P. A.; Giguère, D. Fluorine effect in nucleophilic fluorination at C4 of 1,6-anhydro-2,3-dideoxy-2,3-difluoro-beta-D-hexopyranose. Beilstein J. Org. Chem. 2020, 16, 2880-2887. Part of the thematic issue « Organo-fluorine chemistry V », Guest Editor: D. O’Hagan.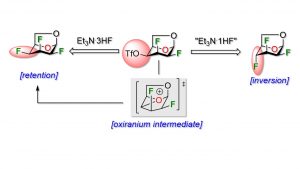
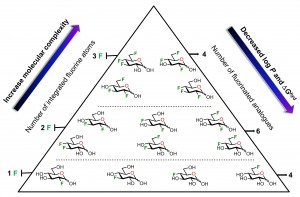
30) St-Gelais, J.; Côté, É.; Lainé, D.; Johnson, P. A.; Giguère, D. Addressing the molecular complexity of fluorinated glucose analogues: Insight into lipophilicities and solvation effects. Chem. Eur. J. 2020, 26, 13499-13506. Also appears in Hot Topics: Fluorine Chemistry.
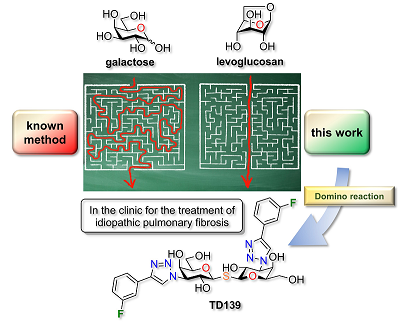
29) St-Gelais, J.; Denavit, V.; Giguère, D. Efficient synthesis of a galectin inhibitor clinical candidate (TD139) using a Payne rearrangement/azidation reaction cascade. Org. Biomol. Chem. 2020, 18, 3903-3907.
28) Tremblay, T.; Robert-Scott, G.; Bérubé, C.; Carpentier, A.; Voyer, N.; Giguère, D. Synthesis of C-terminal glycopeptides via oxime resin aminolysis. Chem. Commun. 2019, 55, 13741-13744.
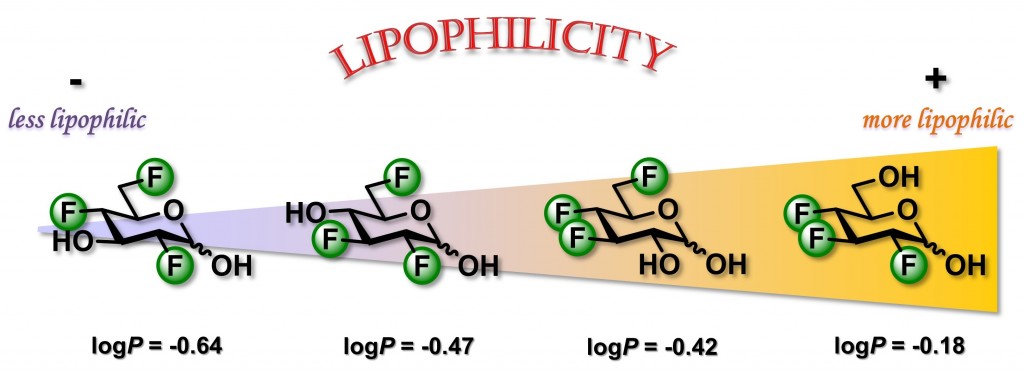
27) St-Gelais, J.; Bouchard, M.; Denavit, V.; Giguere, D. Synthesis and lipophilicity of trifluorinated analogues of glucose. J. Org. Chem. 2019, 84, 8509-8522.
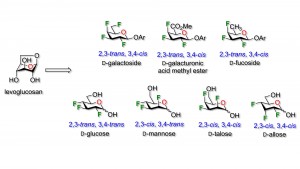
26) Denavit, V.; St-Gelais, J.; Tremblay, T.; Giguere, D. Exploring the chemistry of non-sticky sugars: Synthesis of polyfluorinated carbohydrates analogues of D-allopyranose. Chem. Eur. J. 2019, 25, 9272-9279.
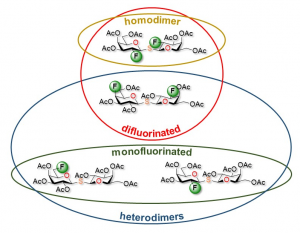
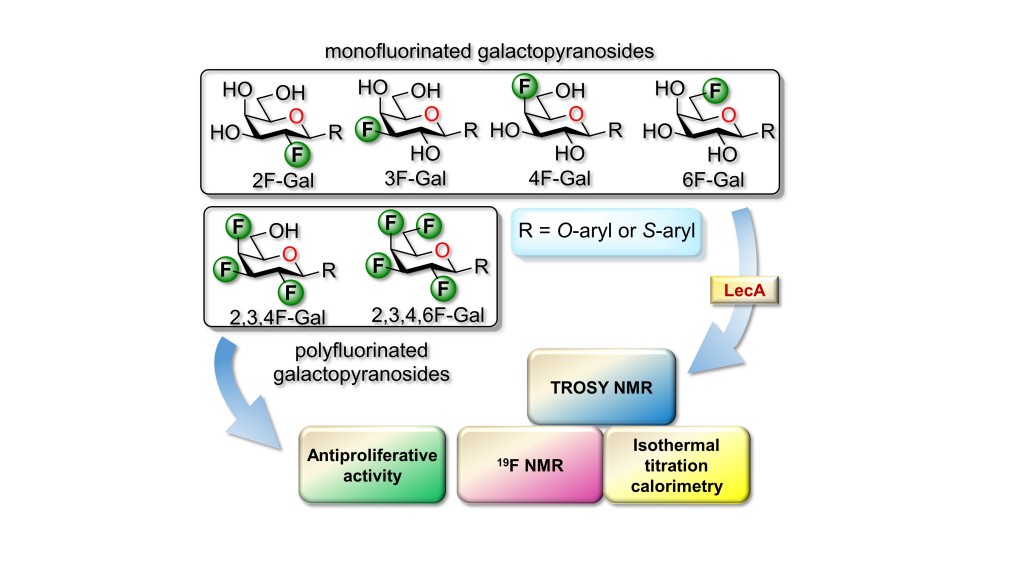
25) Denavit, V.; Laine, D.; Bouzriba, C.; Shanina, E.; Gillon, E.; Fortin, S.; Rademacher, C.; Imberty, A.; Giguere, D. Stereoselective synthesis of fluorinated galactopyranosides as potential molecular probes on galactophilic proteins: assessment of monofluorogalactosides-LecA interactions. Chem. Eur. J. 2019, 25, 4478-4490.
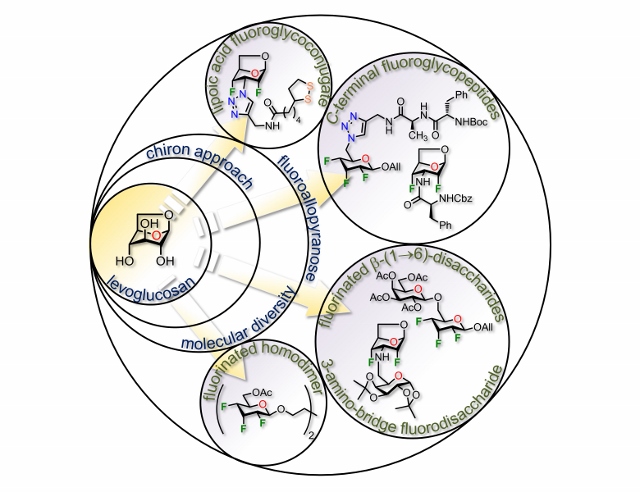
24) Denavit, V.; Laine, D.; St-Gelais, J.; Johnson, P. A.; Giguere, D. A Chiron approach towards the stereoselective synthesis of polyfluorinated carbohydrates. Nat. Commun. 2018, 9, 1-11.
23) Vadhadiya, P. M.; Jean, M.-A.; Bouzriba, C.; Tremblay, T.; Lagüe, P.; Fortin, S.; Boukouvalas, J.; Giguere, D. Diversity-oriented synthesis of diol-based peptidomimetics as potential HIV protease inhibitors and antitumor agents. ChemBioChem. 2018, 19, 1779-1791.
22) Denavit, V.; Laine, D.; Tremblay, T.; St-Gelais, J.; Giguere, D. Synthetic inhibitors of Galectins: Structures and Syntheses. Trends Glycosci. Glycotechnol. 2018, 30, SE21-SE40. Special issue on Galectins.
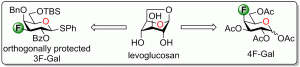
21) Laine, D.; Denavit, V.; Giguere, D. Synthesis of protected 3-deoxy-3-fluoro- and 4-deoxy-4-fluoro-D-galactopyranosides from levoglucosan. J. Org. Chem. 2017, 82, 4986-4992.
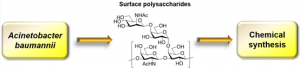
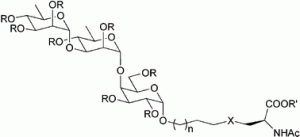
20) Giguere, D. Surface polysaccharides from Acinetobacter baumannii: Structures and syntheses. Carbohydr. Res. 2015, 418, 29-43.
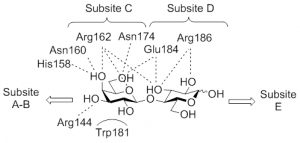
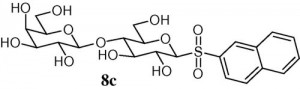
19) Rodrigue, J.; Ganne, G.; Blanchard, B.; Saucier, C.; Giguere, D.; Shiao, T. C.; Varrot, A.; Imberty, A.; Roy, R. Aromatic thioglycoside inhibitors against virulence factor LecA from Pseudomonas aeruginosa. Org. Biomol. Chem. 2013, 11, 6906-6918.
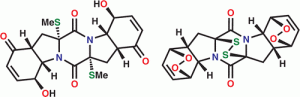
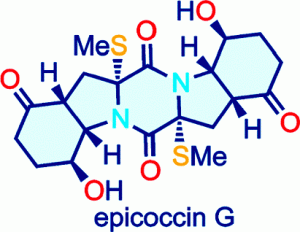
18) Nicolaou, K. C.; Lu, M.; Totokotsopoulos, S.; Heretsch, P.; Giguere, D.; Sun, Y.-P.; Sarlah, D.; Nguyen, T. H.; Wolf, I. C.; Smee, D. F.; Day, C. W.; Bopp, S.; Winzeler, E. A. Synthesis and biological evaluation of epidithio-, epitetrathio-, and bis-(methylthio)diketopiperazines: Synthetic methodology, enantioselective total synthesis of Epicoccin G, 8,8’-epi-ent-Rostratin B, Gliotoxin, Gliotoxin G, Emethallicin E, and Haematocin and discovery of new antiviral and antimalarial agents. J. Am. Chem. Soc. 2012, 134, 17320-17332.
17) Bergeron-Brlek, M.; Giguere, D.; Shiao, T. C.; Saucier, C.; Roy, R. Palladium-catalyzed Ullmann-type reductive homocoupling of iodoaryl glycosides. J. Org. Chem. 2012, 77, 2971-2977.
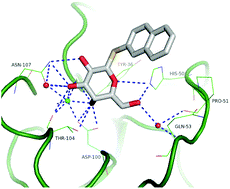
16) St-Pierre, C.; Ouellet, M.; Giguere, D.; Ohtake, R.; Roy, R.; Sato, S. Galectin-1 specific inhibitors as a new class of compounds to treat HIV-1 infection. Antimicrob. Agents Chemother. 2012, 56, 154-162.
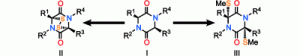
15) Nicolaou, K. C.; Giguere, D.; Totokotsopoulos, S.; Sun, Y.-P. A practical sulfenylation of 2,5-diketopiperazines. Angew. Chem. Int. Ed. 2011, 51, 728-732.
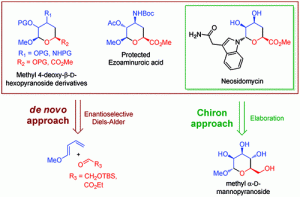
14) Giguere, D.; Martel, J.; Shiao, T. C.; Roy, R. Enantioselective de novo synthesis of 4-deoxy-hexopyranoses via hetero-Diels-Alder cycloadditions-Total synthesis of ezoaminuroic acid and neosidomycin. J. Org. chem. 2011, 76, 9687-9698.
13) Nicolaou, K. C.; Totokotsopoulos, S.; Giguere, D.; Sun, Y.-P.; Sarlah, D. Total synthesis of epicoccin G. J. Am. Chem. Soc. 2011, 133, 8150-8153.
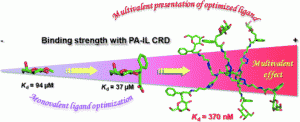
12) Chabre, Y. M.; Giguere, D.; Blanchard, B.; Rodrigue, J.; Rocheleau, S.; Neault, M.; Rauthu, S.; Papadopoulos, A.; Arnold, A.; Imberty, A.; Roy, R. Combining glycomimetic and multivalent strategies toward designing potent bacterial lectin inhibitors. Chem. Eur. J. 2011, 17, 6545-6562.
11) Giguere, D.; Andre, S.; Bonin, M.-A.; Bellefleur, M.-A.; Provencal, A.; Cloutier, P.; Pucci, B.; Roy, R.; Gabius, H.-J. Inhibitory potential of chemical substitutions at bioinspired sites of b-D-galactose on neoglycoprotein/cell surface binding of two classes of medically relevant lectins. Bioorg. Med. Chem. 2011, 19, 3280-3287.
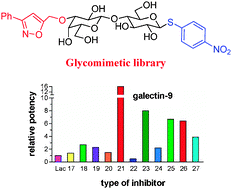
10) Andre, S.; Giguere, D.; Dam, T. K.; Brewer, F.; Gabius, H.-J.; Roy, R. Synthesis of a small glyco-mimetic library for inhibitory activity on medically relevant galactose-specific lectins in assays of increasing bio-relevance. N. J. Chem. 2010, 34, 2229-2240.
9) Giguere, D.; Cloutier, P.; Roy, R. Domino Heck/lactonization-catalyzed synthesis of 3-C-linked mannopyranosyl coumarins. J. Org. Chem. 2009, 74, 8480-8484.
8) Giguere, D.; Patnam, R.; Juarez-Ruiz, J. M.; Neault, M.; Roy, R. Catalytic synthesis of novel 4-C-glycosyl coumarins using a domino Heck reaction/lactonization process. Tetrahedron Lett. 2009, 50, 4254-4257.
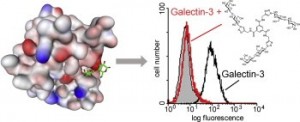
7) Giguere, D.; Bonin, M.-A.; Cloutier, P.; Patnam, R.; St-Pierre, C.; Sato, S.; Roy, R. Synthesis of stable inhibitors of human galectin-1 and -3. Bioorg. Med. Chem. 2008, 16, 7811-7823.
6) Faure, R.; Shiao, T. C.; Lagnoux, D.; Giguere, D.; Roy, R. En route to a carbohydrate-based vaccine against Burkholderia cepacia. Org. Biomol. Chem. 2007, 5, 2704-2708.
5) Bonin, M.-A.; Giguere, D.; Roy, R. N-Arylimidazole synthesis by cross-cycloaddition of isocyanides using a novel catalytic system. Tetrahedron 2007, 63, 4912-4917.
4) Sirois, S.; Giguere, D.; Roy, R. A first QSAR model for galectin-3 glycomimetic inhibitors based on 3D docked structures. Med. Chem. 2006, 2, 481-489.
3) Beha, S.; Giguere, D.; Patnam, R.; Roy, R. Formation of isoxazoles using cerium ammonium nitrate (CAN): a one-pot synthesis of glycomimetics. Synlett 2006, 11, 1739-1743.
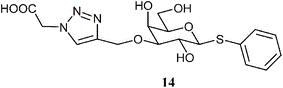
2) Giguere, D.; Patnam, R.; Bellefleur, M.-A.; St-Pierre, C.; Sato, S.; Roy, R. Carbohydrate triazoles and isoxazoles as inhibitors of galectins-1 and -3. Chem. Commun. 2006, 22, 2379-2381.
1) Giguere, D.; Sato, S.; St-Pierre, C.; Sirois, S.; Roy, R. Aryl O- and S-galactosides and lactosides as specific inhibitors of human galectins-1 and -3: Role of electrostatic potential at O-3. Bioorg. Med. Chem. Lett. 2006, 16, 1668-1672.